The pharmaceutical industry lives in a regulated world. In order to comply with the rules of the regulated world, it is always good to check that everyone is also doing according to the rules and instructions. Inspections are conducted by the authorities and audits are usually conducted by external parties outside the company’s pharmacovigilance system. The legislatives and regulations have not been referred here, because they can all be found from the internet.
The authorities publish information on what kind of findings are found in the pharmacovigilance inspections. According to the report published on September 4, 2023 by the UK authority, MHRA, from April 2021 to March 2022, MHRA conducted 32 inspections of pharmacovigilance operations. In these inspections, there were six critical, 72 major and 91 minor findings.
According to the report, the findings of the inspections were divided as follows:
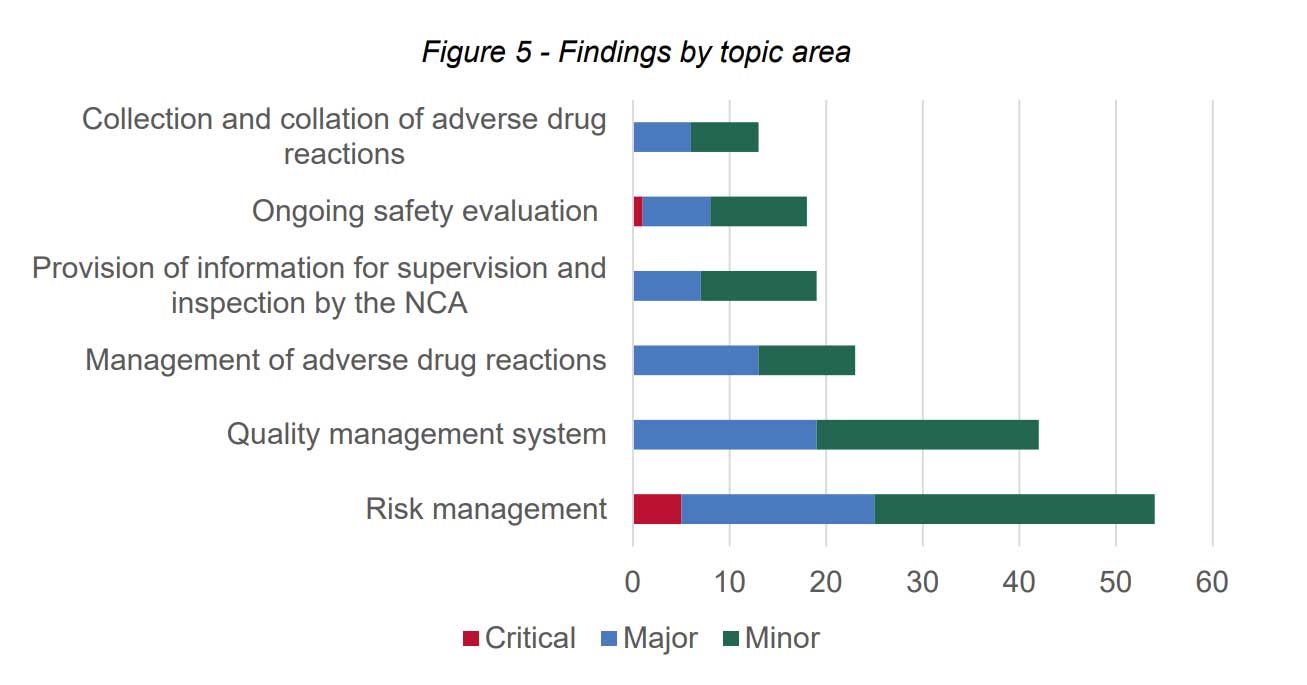
According to a few examples, the critical findings of risk management were
- Due to the nature of the warnings and the medical significance of the safety information that was missing from the PIL, there was an impact on the safety and well-being of patients.
- The continued failure to ensure that the PIL in product packs and on the company website was up-to-date with the current scientific knowledge meant that patients and prescribers did not have the full and comprehensive information available to allow the safe use of the product.
The European Medicines Agency (EMA) publishes an annual report that includes the number of EMA’s pharmacovigilance inspections.
Inspections and audits are a good opportunity to reflect on your own pharmacovigilance activities and think about what is challenging, what is good, and what needs to be developed.
References:
Medicines and Healthcare products Regulatory Agency: Pharmacovigilance Inspection Metrics Report April 2021 – March 2022
Assessed 20 Oct 2023 https://assets.publishing.service.gov.uk/government/uploads/system/uploads/attachment_data/file/1182091/MHRA_GPvP_inspection_metrics_2021-22.pdf
European Medicines Agency: Annual Report 2022
Assessed 20 Oct 2023 https://www.ema.europa.eu/en/annual-report/2022/inspections-and-compliance.html


